  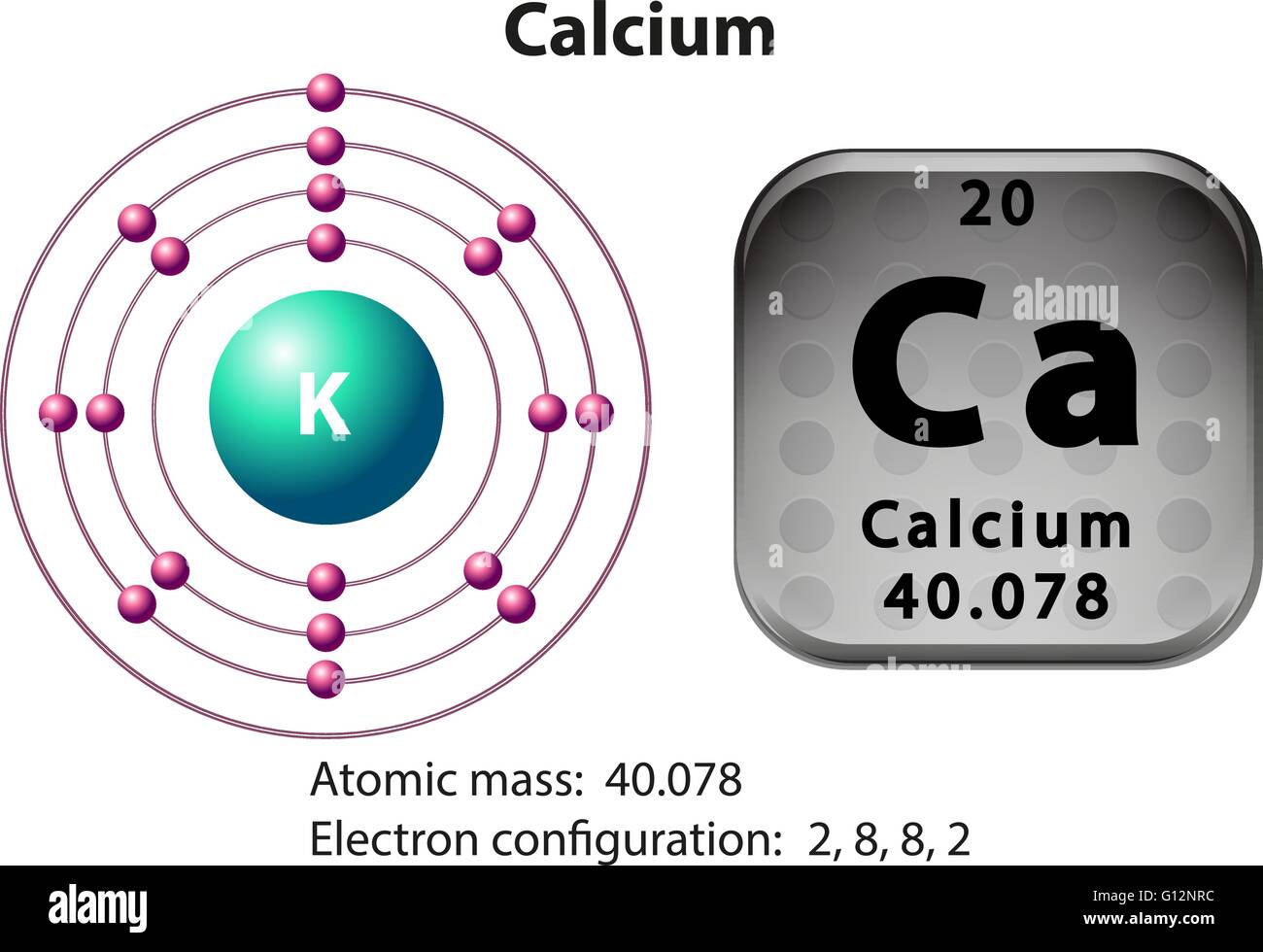
From it, he removed the mercury by distillation and obtained pure calcium. Then he added more lime to the mixture producing a greater amount of the amalgam. Though it did produce an amalgam each of calcium and mercury, the quantity was not enough to establish the existence of a new element. Next, he tried to electrolyze a mixture of lime and mercury oxide. In 1808, Davy tried to reduce moist lime by the process of electrolysis but found no success. However, he suspected that it could be the oxide of an unknown element. French chemist, Antoine Lavoisier classified lime as an ‘earth’ since it seemed impossible to reduce it any more. įor centuries plaster and mortar were made by heating limestone into lime (calcium oxide). How many neutrons are there in its nucleus An atom consists of a nucleus. When Was It Discovered and Where: It was discovered in 1808 in London. An atom of calcium has a mass number of 42 and an atomic number of 20. Who Discovered Calcium: Cornish chemist, Sir Humphry Davy. Origin of Its Name: Its name is derived from the Latin word “calx” meaning lime (limestone is an ore of Ca). It is highly reactive towards the halogens chlorine, bromine, iodine and fluorine to form calcium dihalides. When heated in air, it burns to produce a mixture of calcium oxide and calcium nitride. The surface of the metal remains covered by a thin layer of its oxide that prevents further attack by air (oxygen). It slowly reacts with water to form calcium hydroxide and hydrogen gas. Besides that, it has 19 synthetic, radioactive isotopes with known half-lives. Naturally occurring Ca is a mixture of 6 isotopes with mass numbers 40, 42, 43, 44, 46 and 48. Properties and Characteristics of CalciumĬalcium (pronunciation KAL-see-em ), represented by the chemical symbol or formula Ca, is a soft element belonging to the family of alkali earth metals.It has a melting point of 842°c and a boiling point of 1484°c. It is located in Group 2 as an Alkaline earth metal which is very reactive with water. It is a reactive metal so is not found naturally in the earth’s crust, instead existing as a compound most commonly Calcium Carbonate (Limestone). Calcium is one of the most popular elements in the periodic table because of the abundance of chemical compounds that one can find in nature. Calcium is also used in the paper industry as a bleach and in electrical insulators. In the human body Calcium and Vitamin D interact to improve the uptake of Calcium into bones. A deficiency of calcium can lead to severe diseases such as osteoporosis and osteomalacia. It has a wide variety of uses in the food industry as a supplement for the human body where it is important in nerve transmission and bone strength. Calcium compounds have been known for years from the early days of sculpture using Calcium Carbonate (Limestone) but it was not until 1808 that Sir Humphry Davy isolated Calcium using electrolysis on a mixture of Calcium oxide and Mercuric Oxide. It has the symbol Ca.Ĭalcium is an Alkaline earth metal in Group 2 of the periodic table that is the fifth most abundant element in the earths crust, forming compounds such as limestone and fossils. It is an Alkaline Earth Metal and is located in Group 2 of the periodic table. Calcium (Ca) is a silvery-white soft metal that has the atomic number 20 in the periodic table.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
